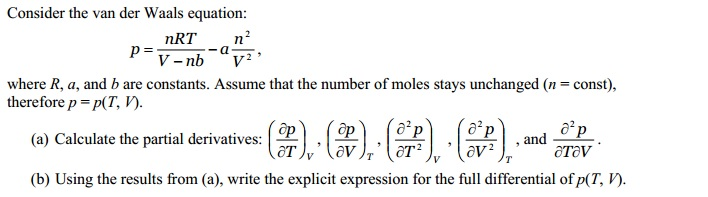
The magnitude of intermolecular attractive forces between the particles is measured by the constant "a." The constant "b" measurement of a gas molecule's volume. In 1873, while searching for a way to link the behavior of liquids and gases, the. At low temperatures or high pressures, real gases deviate significantly from ideal gas behavior. What do the constants A and B in the Van Der Waal Equation mean? The behavior of real gases usually agrees with the predictions of the ideal gas equation to within 5 at normal temperatures and pressures. It also takes into account two other factors: intermolecular attraction and the molecule's finite size.Ĥ. The Van Der Waals equation establishes a link between the volume, pressure, and absolute temperature of a real gas. What Does the Van Der Waals Equation Mean? 
For a dimensional formula is the same as for.ģ. The molar gas constant is R, while the van der Waal constants are a and b. (P+V2a)(V−b)=RT is the van der Waal's gas equation, where P is the pressure, V is the molar volume, and T is the temperature of the provided gas sample. 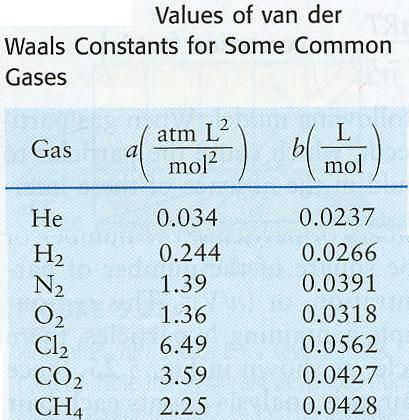
How are the Van Der Waals Constants A and B calculated? The van der Waals equation is a state equation that accounts for two aspects of real gases: gas-particle excluded volume and gas molecule attractive forces. What is the formula for calculating Van Der Waals? The Van Der Waals Equation is an excellent approximation of the genuine gas state equation in most cases, especially at high pressures and around the gas condensation parameters in temperature and pressure.ġ. As a result, we must apply the constraint V / n > b to avoid a situation in which the volume of the molecules exceeds that of the entire gas. The volume of one mole of molecules corresponds to the constant b value (it is a correction for finite molecular size). With the following relationship, you may learn what is the relationship between volume, pressure, and temperature in real gas in the second section of the van der Waals equation calculator i.e. In advanced mode, you can alter those constants directly or select one of the common gases. Van der Waals law calculator is a powerful online tool for solving problems using Van der Waals law equation. To estimate van der Waals constants, you must first provide the crucial parameters of the considered gas. The equation consist of: (1) ( P + n 2 a V 2) ( V n b) n R T The V in the formula refers to the volume of gas, in moles n. The pressure predicted by the van der Waals equation is 21.4 bar.There are two elements to our van der Waals equation calculator. Van der Waals Equation Van der Waals equation is required for special cases, such as non-ideal (real) gases, which is used to calculate an actual value.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
